
Fluid forces are also affected by a number of cardiac cells that were developing into blood cells, lodged in the trabeculation fluid spaces. We also find that there is a squeezing motion of cardiac tissues on the trabeculation fluid spaces, which is the main mechanism that generated fluid forces. We find that they cause fluid forces to have spatial variability and an oscillatory nature. The heart’s inner surface has uneven trabeculation structures. We find that the use of a particular fish line that expresses fluorescence at the exact boundary between heart tissue and blood, that is the endocardial cell membrane boundary, is important to give high quality results. 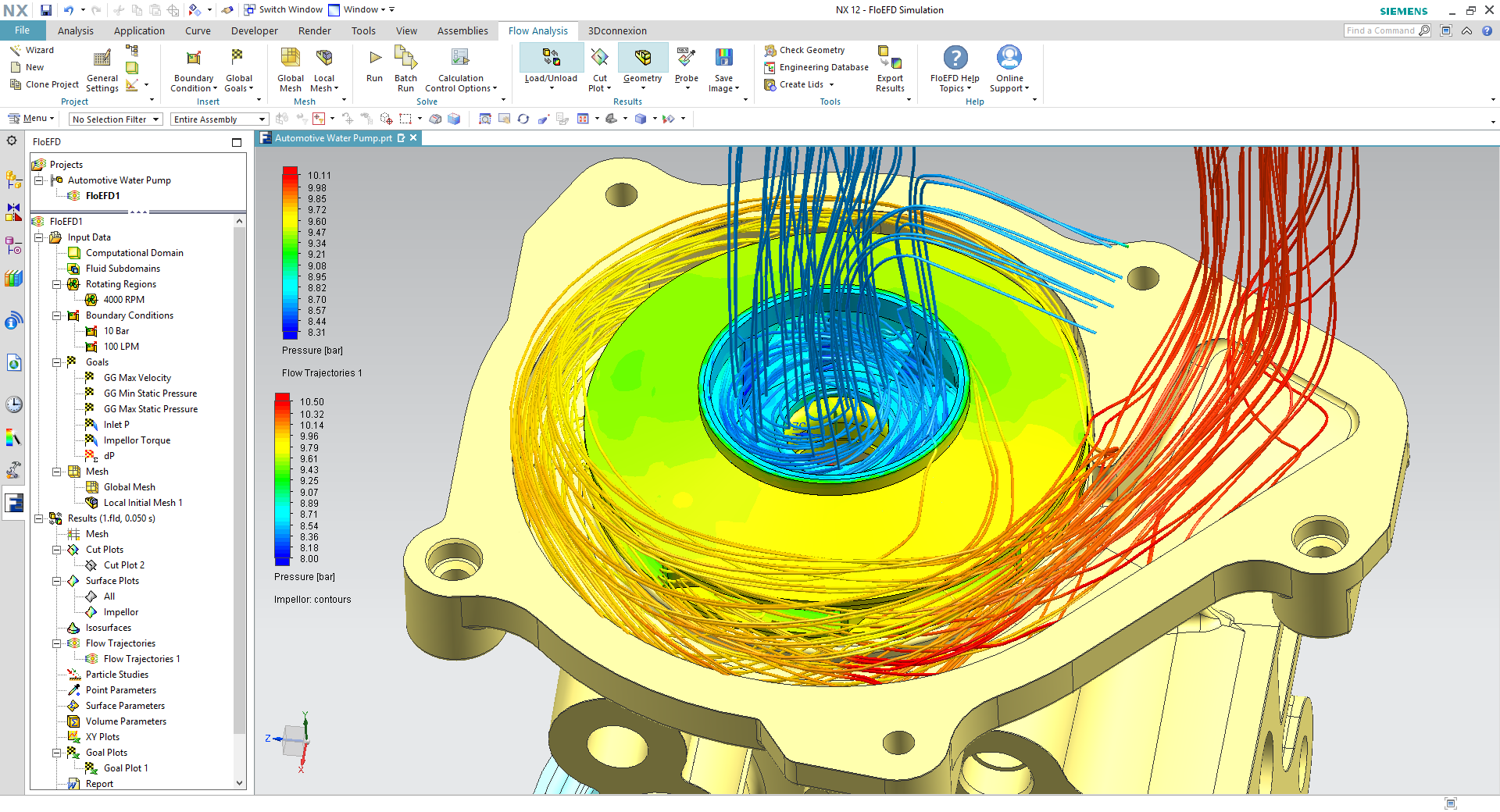 
To do this, we perform high resolution imaging of zebrafish embryonic hearts and image-based flow simulations. We thus perform careful quantification of these forces, using the zebrafish embryo as a model. In the embryonic heart, the mechanical forces that blood fluid imposes on the cardiac tissues are known to be important biological stimuli that affect the proper heart development. Overall, our results suggested that a complex multi-component consideration of both anatomic features and motion dynamics were needed to quantify the trabeculated embryonic heart fluid mechanics. We further found cells attached to the endocardium within the intra-trabecular spaces, which were likely embryonic hemogenic cells, whose presence increased endocardial WSS. We found that trabecular structures were responsible for the high spatial variability of the magnitude and oscillatory nature of WSS, and for reducing the endocardial deformational burden. By conducting simulations of single intra-trabecular spaces under varied scenarios, where the translational or deformational motions (caused by contraction) were removed, we found that a squeeze flow effect was responsible for most of the WSS magnitude in the intra-trabecular spaces, rather than the shear interaction with the flow in the main ventricular chamber. Our endocardial wall shear stress (WSS) results were found to exceed those reported in existing literature, which were estimated using myocardial rather than endocardial boundaries. To capture trabecular geometric and motion details, we used a fish line that expresses fluorescence at the endocardial cell membrane, and high resolution 3D confocal microscopy. Here, we conducted image-based computational fluid dynamics (CFD) simulations to quantify the fluid mechanics associated with the zebrafish embryonic heart trabeculae. However, the fluid dynamical behaviour close to the embryonic endocardial surface is very sensitive to the geometry and motion dynamics of fine-scale cardiac trabecular surface structures. It is thus important to understand the nature of embryonic heart fluid forces. Embryonic heart development is a mechanosensitive process, where specific fluid forces are needed for the correct development, and abnormal mechanical stimuli can lead to malformations.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
